 
For n 1 = 1 the Lyman series is obtained this series lies in the extreme ultraviolet regions of the spectrum n 1 = 2 gives the Balmer series, the lines of which are located in the visible and near ultraviolet regions. defines the single lines of a given series R is the Rydberg constant (expressed in wave numbers). defines a series, and the value n 2 = n 1 + 1, n 1+2, n 1 +3. Where n 1 and n 2 are the values of the main quantum number for energy levels between which a quantum transition occurs. The wave numbers of the lines of its spectrum are defined with extreme accuracy by Balmer’s formula The simplest spectrum is that of the hydrogen atom. The intervals between the lines in a series decrease toward the shortest wavelengths, and the lines converge toward the upper limit of the series. 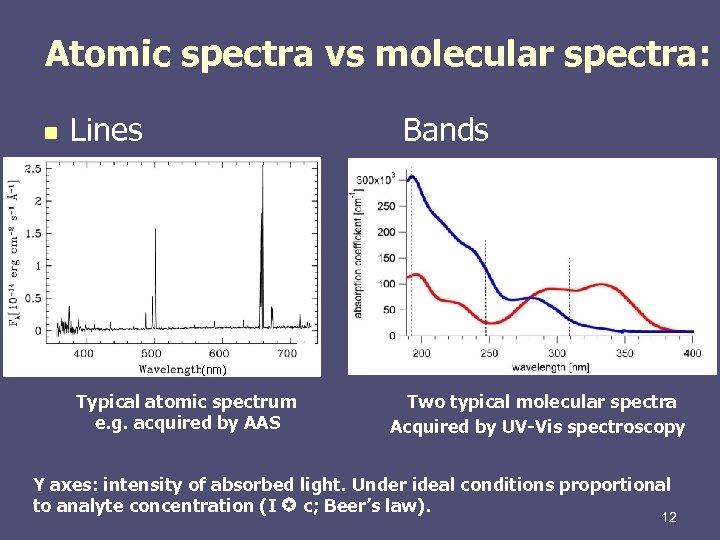
The lines of atomic spectra form regular groups called spectral series. In actual observed spectra, the lines of both neutral and ionized atoms are often present-for instance, the lines Fel, Fell, and Felll in the spectrum of iron, corresponding to Fe, Fe +, and Fe 2+. The spectra of a neutral atom and its sequential ions are signi[ed in spectroscopy by the symbols I, II, III. This displacement is greater the higher the multiplicity of ionization of the atom-the more electrons the atom has lost. (Neutral atoms are easily excited and yield emission spectra in electric arc discharges, but positive ions are less easily excited and yield emission spectra essentially through electrical spark discharge.) The spectra of ionized atoms are displaced with respect to the spectra of neutral atoms into high frequency regions-that is, into the ultraviolet regions. Such spectra are exhibited by neutral as well as ionized atoms they are frequently called arc and spark spectra respectively. 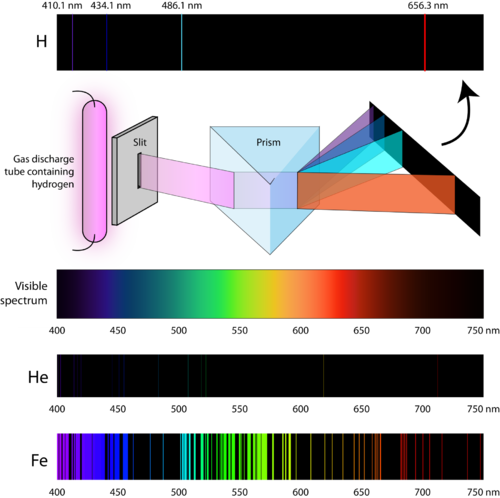
In addition to frequency, the spectral line can also be characterized by wavelength λ = c/v, wave number l/λ = v/c ( c is the velocity of light), and energy of a photon hv.Ītomic spectra arise owing to transitions between energy levels of outer-shell electrons of an atom and are observed in the visible, ultraviolet, and near infrared regions. Each spectral line is characterized by a specific vibration frequency v of emission or absorption of light and pertains to a specific quantum transition between energy levels E 1 and E k of an atom in accordance with the relationship hv = E i - E k (where h is Planck’s constant). They are observed in the form of bright colored lines, resulting from radiation by gases or vapors through electrical arcs or discharges (emission spectra), and in the form of dark lines (absorption spectra). Atomic spectra are linear-they consist of spaced spectral lines. Optical spectra obtained through the emission or absorption of light (electromagnetic waves) by free or weakly bound atoms in particular, monatomic gases and vapors have these spectra. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
